Dosage Forms
Excipient information presented when available (limited, particularly for generics); consult specific product labeling.
Tablet, Oral:
Casodex: 50 mg
Generic: 50 mg
Pharmacology
Mechanism of Action
Bicalutamide is a pure nonsteroidal androgen receptor inhibitor, specifically a competitive inhibitor for the binding of dihydrotestosterone and testosterone; prevents testosterone stimulation of cell growth in prostate cancer
Pharmacokinetics/Pharmacodynamics
Absorption
Well absorbed; unaffected by food
Metabolism
Extensively hepatic; glucuronidation and oxidation of the R (active) enantiomer to inactive metabolites; the S enantiomer is inactive
Excretion
Urine and feces
Time to Peak
Active enantiomer: ~31 hours
Half-Life Elimination
Active enantiomer: ~6 days (~10 days in severe hepatic impairment)
Protein Binding
96%
Use in Specific Populations
Special Populations: Hepatic Function Impairment
The half-life is increased ~76% (to ~10 days) in patients with severe liver disease.
Use: Labeled Indications
Prostate cancer, metastatic: Treatment of stage D2 metastatic prostate cancer (in combination with an LHRH agonist)
Limitation of use: Bicalutamide 150 mg daily is not approved for use alone or with other treatments
Use: Off Label
Locally advanced prostate cancer (monotherapy)b
Data from a large randomized, double blind, placebo-controlled study in patients with prostate cancer suggests a benefit from the use of bicalutamide monotherapy in men with locally-advanced prostate cancer who are at higher risk for recurrence, although men at low risk for recurrence may not benefit from bicalutamide monotherapy McLeod 2006.
Contraindications
Hypersensitivity to bicalutamide or any component of the formulation; use in women; pregnancy
Canadian labeling: Additional contraindications (not in the US labeling): Patients with localized prostate cancer undergoing watchful waiting; children
Dosage and Administration
Dosing: Adult
Prostate cancer, metastatic: Oral: 50 mg once daily (in combination with an LHRH analogue)
Prostate cancer, locally advanced, high recurrence risk (off-label use): Oral: 150 mg once daily (as monotherapy) (McLeod 2006).
Missed doses: If a dose is missed, skip the missed dose and take the next dose at the next scheduled time; do not double the next dose.
Dosing: Geriatric
Refer to adult dosing.
Administration
Oral: Dose should be taken at the same time each day, either in the morning or in the evening. May be administered with or without food. Treatment for metastatic cancer should be started concomitantly with an LHRH analogue.
Storage
Store at room temperature of 20°C to 25°C (68°F to 77°F).
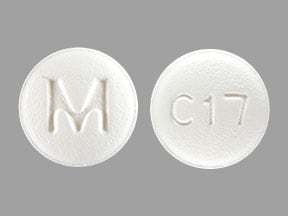
Bicalutamide Images
Drug Interactions
Aminolevulinic Acid (Systemic): Photosensitizing Agents may enhance the photosensitizing effect of Aminolevulinic Acid (Systemic). Avoid combination
Aminolevulinic Acid (Topical): Photosensitizing Agents may enhance the photosensitizing effect of Aminolevulinic Acid (Topical). Monitor therapy
ARIPiprazole: CYP3A4 Inhibitors (Weak) may increase the serum concentration of ARIPiprazole. Management: Monitor for increased aripiprazole pharmacologic effects. Aripiprazole dose adjustments may or may not be required based on concomitant therapy and/or indication. Consult full interaction monograph for specific recommendations. Monitor therapy
Astemizole: Bicalutamide may increase the serum concentration of Astemizole. Avoid combination
Choline C 11: Antiandrogens may diminish the therapeutic effect of Choline C 11. Monitor therapy
Cisapride: Bicalutamide may increase the serum concentration of Cisapride. Avoid combination
Dofetilide: CYP3A4 Inhibitors (Weak) may increase the serum concentration of Dofetilide. Monitor therapy
Flibanserin: CYP3A4 Inhibitors (Weak) may increase the serum concentration of Flibanserin. Monitor therapy
Indium 111 Capromab Pendetide: Antiandrogens may diminish the diagnostic effect of Indium 111 Capromab Pendetide. Avoid combination
Lemborexant: CYP3A4 Inhibitors (Weak) may increase the serum concentration of Lemborexant. Management: The maximum recommended dosage of lemborexant is 5 mg, no more than once per night, when coadministered with weak CYP3A4 inhibitors. Consider therapy modification
Lomitapide: CYP3A4 Inhibitors (Weak) may increase the serum concentration of Lomitapide. Management: Patients on lomitapide 5 mg/day may continue that dose. Patients taking lomitapide 10 mg/day or more should decrease the lomitapide dose by half. The lomitapide dose may then be titrated up to a max adult dose of 30 mg/day. Consider therapy modification
NiMODipine: CYP3A4 Inhibitors (Weak) may increase the serum concentration of NiMODipine. Monitor therapy
Pimozide: CYP3A4 Inhibitors (Weak) may increase the serum concentration of Pimozide. Avoid combination
Porfimer: Photosensitizing Agents may enhance the photosensitizing effect of Porfimer. Monitor therapy
Terfenadine: Bicalutamide may increase the serum concentration of Terfenadine. Avoid combination
Triazolam: CYP3A4 Inhibitors (Weak) may increase the serum concentration of Triazolam. Management: Consider triazolam dose reduction in patients receiving concomitant weak CYP3A4 inhibitors. Consider therapy modification
Ubrogepant: CYP3A4 Inhibitors (Weak) may increase the serum concentration of Ubrogepant. Management: In patients taking weak CYP3A4 inhibitors, the initial and second dose (if needed) of ubrogepant should be limited to 50 mg. Consider therapy modification
Verteporfin: Photosensitizing Agents may enhance the photosensitizing effect of Verteporfin. Monitor therapy
Vitamin K Antagonists (eg, warfarin): Bicalutamide may increase the serum concentration of Vitamin K Antagonists. Specifically, free concentrations of the vitamin K antagonists may be increased. Monitor therapy
Adverse Reactions
Adverse reaction percentages reported as part of combination regimen with an LHRH analogue unless otherwise noted.
>10%:
Cardiovascular: Peripheral edema (13%)
Central nervous system: Pain (35%)
Endocrine & metabolic: Hot flash (53%), gynecomastia (9%; monotherapy [150 mg]: 38% to 73% [McLeod 2006])
Gastrointestinal: Constipation (22%), nausea (15%), diarrhea (12%), abdominal pain (11%)
Genitourinary: Mastalgia (6%; monotherapy [150 mg]: 39% to 85% [McLeod 2006]), pelvic pain (21%), hematuria (12%), nocturia (12%)
Hematologic & oncologic: Anemia (11%)
Infection: Infection (18%)
Neuromuscular & skeletal: Back pain (25%), weakness (22%)
Respiratory: Dyspnea (13%)
≥2% to 10%:
Cardiovascular: Chest pain (8%), hypertension (8%), angina pectoris (2% to <5%), cardiac arrest (2% to <5%), cardiac failure (2% to <5%), coronary artery disease (2% to <5%), edema (2% to <5%), myocardial infarction (2% to <5%), syncope (2% to <5%)
Central nervous system: Dizziness (10%), paresthesia (8%), headache (7%), insomnia (7%), myasthenia (7%), anxiety (5%), chills (2% to <5%), confusion (2% to <5%), drowsiness (2% to <5%), hypertonia (2% to <5%), nervousness (2% to <5%), neuropathy (2% to <5%), depression (4%)
Dermatologic: Skin rash (9%), diaphoresis (6%), alopecia (2% to <5%), pruritus (2% to <5%), xeroderma (2% to <5%)
Endocrine & metabolic: Weight loss (7%), hyperglycemia (6%), weight gain (5%), decreased libido (2% to <5%), dehydration (2% to <5%), gout (2% to <5%), hypercholesterolemia (2% to <5%)
Gastrointestinal: Dyspepsia (7%), anorexia (6%), flatulence (6%), vomiting (6%), dysphagia (2% to <5%), hernia (2% to <5%), melena (2% to <5%), periodontal abscess (2% to <5%), xerostomia (2% to <5%)
Genitourinary: Urinary tract infection (9%), impotence (7%), difficulty in micturition (5%), urinary retention (5%), dysuria (2% to <5%), urinary urgency (2% to <5%), urinary incontinence (4%)
Hematologic & oncologic: Gastrointestinal carcinoma (2% to <5%), rectal hemorrhage (2% to <5%), skin carcinoma (2% to <5%)
Hepatic: Increased liver enzymes (7%), increased serum alkaline phosphatase (5%)
Infection: Herpes zoster (2% to <5%), sepsis (2% to <5%)
Neuromuscular & skeletal: Ostealgia (9%), arthritis (5%), leg cramps (2% to <5%), myalgia (2% to <5%), neck pain (2% to <5%), pathological fracture (4%)
Ophthalmic: Cataract (2% to <5%)
Renal: Polyuria (6%), hydronephrosis (2% to <5%), increased blood urea nitrogen (2% to <5%), increased serum creatinine (2% to <5%)
Respiratory: Cough (8%), pharyngitis (8%), flu-like symptoms (7%), bronchitis (6%), asthma (2% to <5%), epistaxis (2% to <5%), sinusitis (2% to <5%), pneumonia (4%), rhinitis (4%)
Miscellaneous: Cyst (2% to <5%), fever (2% to <5%)
<1%, postmarketing, and/or case reports: Decreased glucose tolerance, decreased hemoglobin, decreased white blood cell count, hepatic failure, hepatitis, hepatotoxicity, hypersensitivity (including angioedema and urticaria), increased serum ALT, increased serum AST, increased serum bilirubin, interstitial pneumonitis, interstitial pulmonary disease (most often at doses >50 mg), pulmonary fibrosis, skin photosensitivity
Warnings/Precautions
Concerns related to adverse effects:
- Gynecomastia: Bicalutamide may cause gynecomastia or breast pain at higher (off-label) doses.
- Hematologic: Anemia may occur with testosterone suppression; monitor CBC periodically as indicated.
- Hepatotoxicity: Cases of death or hospitalization due to severe liver injury have been reported (postmarketing); hepatotoxicity generally occurs within the first 3 to 4 months of treatment. Use with caution in moderate to severe hepatic dysfunction. Monitor transaminases (baseline and periodically); if clinical signs/symptoms suggestive of liver dysfunction (eg, nausea, vomiting, abdominal pain, fatigue, anorexia, flu-like symptoms, dark urine, jaundice, right upper quadrant pain) occur, promptly monitor liver function tests; discontinue if patients have jaundice or ALT is >2 × ULN.
- Hypersensitivity: Angioneurotic edema and urticaria have been reported.
- Interstitial lung disease: Interstitial lung disease has been reported rarely (including fatalities) although mostly at dosages greater than what is recommended; promptly evaluate any worsening of respiratory symptoms (eg, dyspnea, cough and fever).
Disease-related concerns:
- Cardiovascular disease: Androgen-deprivation therapy may increase the risk for cardiovascular disease (Levine 2010). Androgen deprivation therapy may cause prolongation of the QT/QTc interval (Garnick 2004); evaluate risk versus benefit in patients with congenital long QT syndrome, heart failure, frequent electrolyte abnormalities, and in patients taking medication known to prolong the QT interval. Correct electrolytes prior to initiation and consider periodic electrolyte and ECG monitoring.
- Decreased bone mineral density: Prolonged use of antiandrogen therapy is associated with decreased bone mineral density and an increased risk of osteoporosis and fracture (Smith 2003); alcohol abuse, familial history of osteoporosis, and/or chronic use of drugs capable of decreasing bone mass (eg, corticosteroids) may increase risk. Evaluate risk carefully before initiating therapy.
- Diabetes: When used in combination with LHRH agonists, a loss of glycemic control and decrease in glucose tolerance has been reported in patients with diabetes; monitor.
- Hepatic impairment: Use with caution in moderate to severe impairment; bicalutamide undergoes extensive hepatic metabolism. Limited data suggest excretion may be delayed in severe impairment leading to further drug accumulation. Consider periodic monitoring of liver function with prolonged therapy.
Concurrent drug therapy issues:
- Drug-drug interactions: Potentially significant interactions may exist, requiring dose or frequency adjustment, additional monitoring, and/or selection of alternative therapy. Consult drug interactions database for more detailed information.
- Warfarin: Excessive prolongation of prothrombin time (PT) and INR days to weeks after bicalutamide initiation in patients previously stable on warfarin therapy have been reported (postmarketing). Serious bleeding events, including intracranial, retroperitoneal, and GI events (requiring blood transfusion and/or phytonadione [vitamin K]), occurred. Monitor PT/INR in patients on concomitant warfarin therapy, and adjust warfarin dose as necessary.
Other warnings/precautions:
- Anti-androgen withdrawal syndrome: Discontinue use immediately if disease worsens; decreased prostate specific antigen (PSA) levels and/or clinical improvement may be observed in some patients when antiandrogen therapy is held due to worsening of disease.
Monitoring Parameters
Periodically monitor CBC, ECG, echocardiograms, serum testosterone, luteinizing hormone, and prostate specific antigen (PSA). Liver function tests should be obtained at baseline and repeated regularly during the first 4 months of treatment, and periodically thereafter; monitor for signs and symptoms of liver dysfunction. Monitor blood glucose in patients with diabetes. If initiating bicalutamide in patients who are on warfarin, closely monitor prothrombin time and INR. Monitor adherence.
Pregnancy
Pregnancy Considerations
Bicalutamide is contraindicated for use in women, including pregnant women.
Based on the mechanism of action and findings in animal reproduction studies, bicalutamide may cause morphological changes in spermatozoa, inhibit spermatogenesis, and impair male fertility. Males with female partners of reproductive potential should use effective contraception during therapy and for 130 days after the last bicalutamide dose.
Patient Education
What is this drug used for?
- It is used to treat prostate cancer.
- It may be given to you for other reasons. Talk with the doctor.
Frequently reported side effects of this drug
- Nausea
- Diarrhea
- Fatigue
- Anxiety
- Constipation
- Sexual dysfunction
- Hot flashes
- Sensation of warmth
- Sweating a lot
- Back pain
- Abdominal pain
- Pelvic pain
Other side effects of this drug: Talk with your doctor right away if you have any of these signs of:
- High blood sugar like confusion, fatigue, increased thirst, increased hunger, passing a lot of urine, flushing, fast breathing, or breath that smells like fruit
- Infection
- Liver problems like dark urine, fatigue, lack of appetite, nausea, abdominal pain, light-colored stools, vomiting, or yellow skin
- Severe pulmonary disorder like lung or breathing problems like trouble breathing, shortness of breath, or a cough that is new or worse
- Shortness of breath
- Excessive weight gain
- Swelling of arms or legs
- Enlarged breasts
- Breast pain
- Vision changes
- Unable to pass urine
- Change in amount of urine passed
- Chest pain
- Blood in the urine
- Severe dizziness
- Passing out
- Muscle weakness
- Severe loss of strength and energy
- Severe headache
- Signs of a significant reaction like wheezing; chest tightness; fever; itching; bad cough; blue skin color; seizures; or swelling of face, lips, tongue, or throat.
Note: This is not a comprehensive list of all side effects. Talk to your doctor if you have questions.
Consumer Information Use and Disclaimer: This information should not be used to decide whether or not to take this medicine or any other medicine. Only the healthcare provider has the knowledge and training to decide which medicines are right for a specific patient. This information does not endorse any medicine as safe, effective, or approved for treating any patient or health condition. This is only a brief summary of general information about this medicine. It does NOT include all information about the possible uses, directions, warnings, precautions, interactions, adverse effects, or risks that may apply to this medicine. This information is not specific medical advice and does not replace information you receive from the healthcare provider. You must talk with the healthcare provider for complete information about the risks and benefits of using this medicine.