Dosage Forms
Excipient information presented when available (limited, particularly for generics); consult specific product labeling.
Solution, Intravenous:
Boniva: 3 mg/3 mL (3 mL)
Generic: 3 mg/3 mL (3 mL)
Solution, Intravenous [preservative free]:
Generic: 3 mg/3 mL (3 mL)
Tablet, Oral:
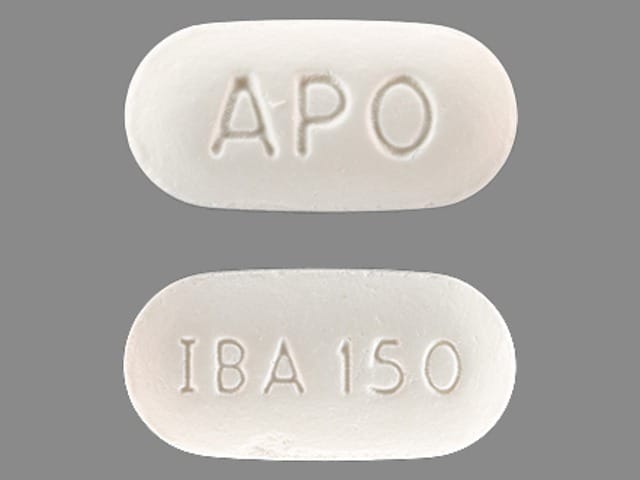
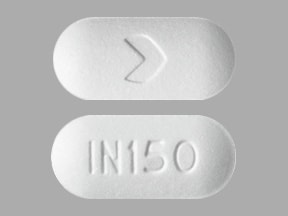
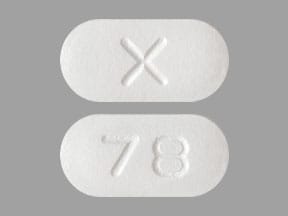
Boniva: 150 mg
Generic: 150 mg
Pharmacology
Mechanism of Action
A bisphosphonate which inhibits bone resorption via actions on osteoclasts or on osteoclast precursors; decreases the rate of bone resorption, leading to an indirect increase in bone mineral density.
Pharmacokinetics/Pharmacodynamics
Distribution
Terminal Vd: 90 L; 40% to 50% of circulating ibandronate binds to bone
Metabolism
Not metabolized
Excretion
Urine (50% to 60% of absorbed dose, excreted as unchanged drug); feces (unabsorbed drug)
Time to Peak
Oral: 0.5 to 2 hours
Half-Life Elimination
Oral: 150 mg dose: Terminal: 37 to 157 hours
IV: Terminal: ∼5 to 25 hours
Protein Binding
85.7% to 99.5%
Use in Specific Populations
Special Populations: Renal Function Impairment
Patients with CrCl 40 to 70 mL/minute had 55% higher AUC and patients with CrCl 30 mL/minute had more than a 2-fold increase in exposure.
Special Populations: Elderly
Progressive age-related changes in renal function may alter the elimination of ibandronate in elderly patients.
Use: Labeled Indications
Osteoporosis: Treatment and prevention of osteoporosis in postmenopausal females. Note: Due to lack of direct evidence for prevention of hip or nonvertebral fracture, ibandronate is not a preferred initial agent compared to other available bisphosphonates; however, initiation in patients requiring vertebral efficacy may be appropriate (AACE [Camacho 2016]; Adler 2016; ES [Eastell 2019]).
Use: Off Label
Breast cancer, metastatic bone disease (treatment)a
Data (including long-term follow up) from a phase III, double-blind, randomized, placebo-controlled trial support the use of ibandronate in the management of symptoms of bone disease due to metastatic breast cancer Body 2003, Diel 2004, Pecherstorfer 2006.
Hypercalcemia of malignancyb
Data from a multicenter, double-blind, randomized dose-comparison study and an open-label, randomized trial support the use of ibandronate in the management of hypercalcemia of malignancy Pecherstorfer 2003, Ralston 1997.
Prostate cancer, metastatic, bone pain (alternative agent)c
A randomized controlled study that demonstrated similar outcomes when comparing ibandronate to radiation therapy for the management of metastatic bone pain suggests that ibandronate may be a treatment alternative for bone pain in patients with metastatic prostate cancer when radiation therapy is not an option Hoskin 2015.
Contraindications
Known hypersensitivity to ibandronate or any component of the formulation; hypocalcemia; oral tablets are also contraindicated in patients unable to stand or sit upright for at least 60 minutes and in patients with abnormalities of the esophagus which delay esophageal emptying, such as stricture or achalasia.
Documentation of allergenic cross-reactivity for bisphosphonates is limited. However, because of similarities in chemical structure and/or pharmacologic actions, the possibility of cross-sensitivity cannot be ruled out with certainty.
Dosage and Administration
Dosing: Adult
Note: Patients treated for osteoporosis should receive supplemental calcium and vitamin D if dietary intake is inadequate.
Breast cancer, metastatic bone disease (treatment; off-label use): IV: 6 mg over 1 to 2 hours every 3 to 4 weeks for up to 4 years (Body 2003; Diel 2004; Pecherstorfer 2006).
Hypercalcemia of malignancy (off-label use): IV: 2 to 6 mg as a single dose over 1 to 2 hours (Pecherstorfer 2003; Ralston 1997).
Osteoporosis, prevention of fractures (postmenopausal females) (alternative agent): Note: Due to lack of direct evidence for prevention of hip or nonvertebral fractures, ibandronate is generally not a preferred bisphosphonate (AACE [Camacho 2016]; ES [Eastell 2019]).
High fracture risk patients, including those with a history of fragility fracture, or postmenopausal females with a T-score ≤−2.5, or a T-score between −1 and −2.5 at high fracture risk according to an assessment (ES [Eastell 2019]; NOF [Cosman 2014]; Siris 2014):
Treatment:
Oral: 150 mg once monthly.
IV: 3 mg every 3 months.
Patients with T-scores between −1 and −2.5 and not at high fracture risk; patient preferences may guide decision to initiate therapy (NOF [Cosman 2014]):
Prevention: Oral: 150 mg once monthly.
Duration of therapy: The optimal duration of therapy has not been established. If fracture risk remains high (eg, fragility fracture before or during therapy) after the initial 3 years (IV) or 5 years (oral), consider extending IV bisphosphonate therapy for up to 6 years and oral bisphosphonate therapy for up to 10 years (based on data with other bisphosphonates) or switching to alternative therapy (AACE/ACE [Camacho 2016]; Adler 2016; ES [Eastell 2019]; Watts 2010). Alternatively, if bone mineral density (BMD) is stable, there have been no previous fragility fractures, and short-term fracture risk is low, consider discontinuation (ie, drug holiday) after the initial 3 years (IV) or 5 years (oral). The optimal length of a drug holiday has not been established, although it is usually for a period of up to 5 years (ES [Eastell 2019]). The decision to resume therapy after a drug holiday is based on multiple factors, including decline in BMD and risk factors for fracture (AACE/ACE [Camacho 2016]; Adler 2016; ES [Eastell 2019]).
Prostate cancer, metastatic, bone pain (alternative agent if radiation therapy is not an option; off-label use) IV: 6 mg as a single dose over 15 minutes (Hoskin 2015).
Missed doses:
Oral (once-monthly): If an oral dose is missed, administer the next morning after remembered if the next month's scheduled dose is >7 days away. If the next month's scheduled dose is within 7 days, wait until the next month's scheduled dose. Then return to the original scheduled day of the month on the once-monthly schedule; however, do not administer >150 mg within 7 days.
IV (once every 3 months): If an IV dose is missed, administer as soon as it can be rescheduled. Thereafter, administer every 3 months from the date of the last injection.
Dosing: Geriatric
Refer to adult dosing.
Administration
Oral: Administer 60 minutes before the first food or drink of the day (other than water) and prior to taking any oral medications or supplements (eg, calcium, antacids, vitamins). Ibandronate should be taken in an upright position with a full glass (6 to 8 oz) of plain water and the patient should avoid lying down for 60 minutes to minimize the possibility of GI side effects. Mineral water with a high calcium content should be avoided. The tablet should be swallowed whole; do not chew or suck. Do not eat or drink anything (except water) for 60 minutes following administration of ibandronate.
IV: Administer as a 15 to 30 second bolus IV; avoid paravenous or intraarterial administration (may cause tissue damage). Do not mix with calcium-containing solutions or other drugs. For osteoporosis, do not administer more frequently than every 3 months.
Off-label rates: Infuse over 1 to 2 hours for metastatic bone disease due to breast cancer and for hypercalcemia of malignancy (Body 2003; Pecherstorfer 2003; Ralston 1997). Infuse over 15 minutes for metastatic bone pain due to prostate cancer (Hoskin 2015).
Dietary Considerations
Ensure adequate calcium and vitamin D intake; if dietary intake is inadequate, dietary supplementation is recommended. Males and females should consume:
Calcium: 1,000 mg/day (males: 50 to 70 years) or 1,200 mg/day (females ≥51 years and males ≥71 years) (IOM 2011; NOF [Cosman 2014]).
Vitamin D: 800 to 1,000 int. units daily (males and females ≥50 years) (NOF [Cosman 2014]). Recommended Dietary Allowance (RDA): 600 int. units daily (males and females ≤70 years) or 800 int. units daily (males and females ≥71 years) (IOM 2011).
Ibandronate tablet should be taken with a full glass (6 to 8 oz) of plain water, at least 60 minutes prior to any food, beverages, or medications. Mineral water with a high calcium content should be avoided.
Storage
Store at 25°C (77°F); excursions permitted to 15°C to 30°C (59°F to 86°F).
Ibandronate Images
Drug Interactions
Aminoglycosides: May enhance the hypocalcemic effect of Bisphosphonate Derivatives. Monitor therapy
Angiogenesis Inhibitors (Systemic): May enhance the adverse/toxic effect of Bisphosphonate Derivatives. Specifically, the risk for osteonecrosis of the jaw may be increased. Monitor therapy
Deferasirox: Bisphosphonate Derivatives may enhance the adverse/toxic effect of Deferasirox. Specifically, the risk for GI ulceration/irritation or GI bleeding may be increased. Monitor therapy
Nonsteroidal Anti-Inflammatory Agents: May enhance the adverse/toxic effect of Bisphosphonate Derivatives. Both an increased risk of gastrointestinal ulceration and an increased risk of nephrotoxicity are of concern. Monitor therapy
Polyvalent Cation Containing Products: May decrease the serum concentration of Bisphosphonate Derivatives. Management: Avoid administration of oral medications containing polyvalent cations within: 2 hours before or after tiludronate/clodronate/etidronate; 60 minutes after oral ibandronate; or 30 minutes after alendronate/risedronate. Consider therapy modification
Proton Pump Inhibitors: May diminish the therapeutic effect of Bisphosphonate Derivatives. Monitor therapy
Test Interactions
Bisphosphonates may interfere with diagnostic imaging agents such as technetium-99m-diphosphonate in bone scans.
Adverse Reactions
Percentages vary based on frequency of administration (daily vs monthly). Unless specified, percentages are reported with oral use.
>10%:
Gastrointestinal: Dyspepsia (4% to 12%)
Neuromuscular & skeletal: Back pain (4% to 14%)
Respiratory: Upper respiratory tract infection (2% to 34%)
1% to 10%:
Cardiovascular: Hypertension (6% to 7%)
Central nervous system: Headache (3% to 7%), dizziness (1% to 4%), fatigue (3%), insomnia (1% to 2%), depression (2%)
Dermatologic: Skin rash (1% to 2%)
Gastrointestinal: Abdominal pain (5% to 8%), diarrhea (2% to 7%), nausea (4% to 5%), dental disease (4%), constipation (3% to 4%), vomiting (3%), gastritis (2%), gastroenteritis (3%)
Genitourinary: Urinary tract infection (2% to 6%), cystitis (3%)
Hypersensitivity: Acute phase reaction-like symptoms (IV: 10%; oral: 3% to 9%), hypersensitivity reaction (3%)
Infection: Influenza (4% to 8%)
Local: Injection site reaction (<2%)
Neuromuscular & skeletal: Limb pain (1% to 8%), arthralgia (4% to 9%), myalgia (1% to 6%), arthropathy (4%), weakness (4%), localized osteoarthritis (1% to 3%), muscle cramps (2%)
Respiratory: Bronchitis (3% to 10%), pneumonia (6%), nasopharyngitis (3% to 4%), flu-like symptoms (1% to 3%), pharyngitis (3%)
Postmarketing and/or case reports: Acute renal failure, anaphylactic shock, anaphylaxis, angioedema, bronchospasm, bullous dermatitis, erythema multiforme, exacerbation of asthma, femur fracture (diaphyseal or subtrochanteric), hypocalcemia, iritis, musculoskeletal pain (bone, joint, or muscle; incapacitating), ophthalmic inflammation, osteonecrosis (oro-facial sites including the external auditory canal), osteonecrosis of the jaw, prolonged Q-T interval on ECG (Bonilla 2014), scleritis, Stevens-Johnson syndrome, uveitis
Warnings/Precautions
Concerns related to adverse effects:
- Bone fractures: Atypical femur fractures (AFF) have been reported in patients receiving bisphosphonates. The fractures include subtrochanteric femur (bone just below the hip joint) and diaphyseal femur (long segment of the thigh bone). Some patients experience prodromal pain weeks or months before the fracture occurs. It is unclear if bisphosphonate therapy is the cause for these fractures; AFFs have also been reported in patients not taking bisphosphonates and in patients receiving glucocorticoids. Patients receiving long-term (>3 to 5 years) bisphosphonate therapy may be at an increased risk (Adler 2016; NOF [Cosman 2014]); however, benefits of therapy (when used for osteoporosis) generally outweigh absolute risk of AFF within the first 5 years of treatment, especially in patients with high fracture risk (Adler 2016; ES [Eastell 2019]). Patients presenting with thigh or groin pain with a history of receiving bisphosphonates should be evaluated for femur fracture. Consider interrupting bisphosphonate therapy in patients who develop a femoral shaft fracture; assess for fracture in the contralateral limb.
- Bone/joint/muscle pain: Infrequently, severe (and occasionally debilitating) bone, joint, and/or muscle pain have been reported during bisphosphonate treatment. The onset of pain ranged from a single day to several months. Discontinue intravenous ibandronate therapy in patients who experience severe symptoms; symptoms usually resolve upon discontinuation. Some patients experienced recurrence when rechallenged with the same drug or another bisphosphonate; avoid use in patients with a history of these symptoms in association with bisphosphonate therapy.
- GI mucosa irritation: May cause irritation to upper GI mucosa. Esophagitis, dysphagia, esophageal ulcers, esophageal erosions, and esophageal stricture (rare) have been reported with oral bisphosphonates; risk increases in patients unable to comply with dosing instructions. Use with caution in patients with dysphagia, esophageal disease, gastritis, duodenitis, or ulcers (may worsen underlying condition). Discontinue if new or worsening symptoms develop.
- Hypersensitivity: Allergic reactions, including anaphylactic reaction/shock (some fatal), angioedema, bronchospasm, exacerbation of asthma, rash, Stevens-Johnson syndrome, erythema multiforme, and dermatitis bullous have been reported; discontinue if hypersensitivity reaction occurs.
- Hypocalcemia: Hypocalcemia has been reported with the use of bisphosphonates. Prior to therapy initiation, hypocalcemia must be corrected; ensure adequate calcium and vitamin D intake.
- Influenza-like illness/acute phase reaction: A transient acute phase reaction (eg, fever, chills, pain/myalgia, or other influenza-like symptoms) may occur, typically within 3 days following the initial infusion; resolution is usually observed within 48 hours after symptom onset but may rarely last >7 days (Popp 2017; Sieber 2013).
- Ocular effects: Uveitis and scleritis have been reported with ibandronate; patients presenting with signs of ocular inflammation may require further ophthalmologic evaluation.
- Osteonecrosis of the jaw: Osteonecrosis of the jaw (ONJ), also referred to as medication-related osteonecrosis of the jaw (MRONJ), has been reported in patients receiving bisphosphonates. Known risk factors for MRONJ include invasive dental procedures (eg, tooth extraction, dental implants, boney surgery), cancer diagnosis, concomitant therapy (eg, chemotherapy, corticosteroids, angiogenesis inhibitors), poor oral hygiene, ill-fitting dentures, and comorbid disorders (anemia, coagulopathy, infection, preexisting dental or periodontal disease). Risk may increase with increased duration of bisphosphonate use. According to a position paper by the American Association of Maxillofacial Surgeons (AAOMS), MRONJ has been associated with bisphosphonates and other antiresorptive agents (denosumab), and antiangiogenic agents (eg, bevacizumab, sunitinib) used for the treatment of osteoporosis or malignancy; risk is significantly higher in cancer patients receiving antiresorptive therapy compared to patients receiving osteoporosis treatment (regardless of medication used or dosing schedule). MRONJ risk is also increased with monthly IV antiresorptive therapy compared to the minimal risk associated with oral bisphosphonate use, although risk appears to increase with oral bisphosphonates when duration of therapy exceeds 4 years (AAOMS [Ruggiero 2014]). The manufacturer's labeling states that discontinuing bisphosphonates in patients requiring invasive dental procedures may reduce the risk of ONJ and clinical judgment by physician and/or oral surgeon should be used. However, the AAOMS suggests there is currently no evidence that interrupting oral bisphosphonate therapy alters the risk of ONJ following tooth extraction, and that no alternations or delay in any procedure common to oral/maxillofacial surgeons, periodontists, and other dental providers is necessary in patients receiving oral bisphosphonates for <4 years who have no clinical risk factors (special considerations apply to patients receiving dental implants). Conversely, in patients receiving oral bisphosphonates for >4 years or in patients receiving oral bisphosphonates for <4 years who have also taken corticosteroids or antiangiogenic medications concomitantly, the AAOMS recommends considering a 2-month drug free period prior to invasive dental procedures based on a theoretical benefit. Patients developing ONJ during therapy should receive care by an oral surgeon (AAOMS [Ruggiero 2014]). According to the manufacturer, discontinuation of oral bisphosphonate therapy should be considered (based on risk/benefit evaluation) in patients who develop ONJ.
Disease-related concerns:
- Bariatric surgery: Altered absorption and ulceration risk: Avoid oral bisphosphates after bariatric surgery; inadequate oral absorption and potential anastomotic ulceration may occur. If therapy is indicated, IV administered bisphosphonates are recommended.
- Renal impairment: Use not recommended with severe renal impairment (CrCl <30 mL/minute). Intravenous bisphosphonate use has been associated with renal deterioration, including acute renal failure.
Concurrent drug therapy issues:
- Drug-drug interactions: Potentially significant interactions may exist, requiring dose or frequency adjustment, additional monitoring, and/or selection of alternative therapy. Consult drug interactions database for more detailed information.
Monitoring Parameters
Osteoporosis: Serial bone mineral density (BMD) should be evaluated at baseline and every 1 to 3 years on treatment (usually at ~2 years following initiation of therapy, then more or less frequently depending on patient-specific factors and stability of BMD) (AACE/ACE [Camacho 2016]; ES [Eastell 2019]; NOF [Cosman 2014]); evaluate BMD every 2 to 4 years during a drug holiday (ES [Eastell 2019]); serum creatinine prior to each IV dose; annual measurements of height and weight, assessment of chronic back pain; serum calcium and 25(OH)D; may consider measuring biochemical markers of bone turnover (eg, fasting serum CTX or urinary NTX) at baseline, 3 months, and 6 months, to assess treatment response, adherence to therapy, and/or possible malabsorption (ES [Eastell 2019]).
Pregnancy
Pregnancy Considerations
It is not known if bisphosphonates cross the placenta, but based on their lower molecular weight, fetal exposure is expected (Djokanovic 2008; Stathopoulos 2011).
Information related to the use of ibandronate in pregnancy is limited (El-Safadi 2012).
Bisphosphonates are incorporated into the bone matrix and gradually released over time. The amount available in the systemic circulation varies by drug, dose, and duration of therapy. Theoretically, there may be a risk of fetal harm when pregnancy follows the completion of therapy (hypocalcemia, low birth weight, and decreased gestation have been observed in some case reports); however, available data have not shown that exposure to bisphosphonates during pregnancy significantly increases the risk of adverse fetal events (Djokanovic 2008; Green 2014; Levy 2009; Sokal 2019; Stathopoulos 2011). Exposed infants should be monitored for hypocalcemia after birth (Djokanovic 2008; Stathopoulos 2011).
Until additional data are available, most sources recommend discontinuing bisphosphonate therapy in females of reproductive potential as early as possible prior to a planned pregnancy. Use in premenopausal females should be reserved for special circumstances when rapid bone loss is occurring; a bisphosphonate with the shortest half-life should then be used (Bhalla 2010; Pereira 2012; Stathopoulos 2011).
Patient Education
What is this drug used for?
- It is used to treat soft, brittle bones (osteoporosis) in women after change of life.
- It is used to put off soft, brittle bones (osteoporosis) in women after change of life.
- It may be given for other reasons. Talk with the doctor.
Frequently reported side effects of this drug
- Nausea
- Headache
- Abdominal pain
- Vomiting
- Diarrhea
- Flu-like symptoms
- Back pain
- Painful extremities
- Nose irritation
- Sore throat
Other side effects of this drug: Talk with your doctor right away if you have any of these signs of:
- Low calcium like muscle cramps or spasms, numbness and tingling, or seizures
- Kidney problems like unable to pass urine, blood in the urine, change in amount of urine passed, or weight gain
- Severe injection site redness, burning, pain, swelling, or irritation
- Painful urination
- Eye pain
- Vision changes
- Severe bone pain
- Severe muscle pain
- Severe joint pain
- Groin, hip, or thigh pain
- Severe dizziness
- Passing a lot of urine
- Jaw pain or swelling
- Chest pain
- Heartburn
- Black, tarry, or bloody stools
- Coughing up blood
- Vomiting blood
- Trouble swallowing
- Pain with swallowing
- Mouth sores
- Signs of a significant reaction like wheezing; chest tightness; fever; itching; bad cough; blue skin color; seizures; or swelling of face, lips, tongue, or throat.
Note: This is not a comprehensive list of all side effects. Talk to your doctor if you have questions.
Consumer Information Use and Disclaimer: This information should not be used to decide whether or not to take this medicine or any other medicine. Only the healthcare provider has the knowledge and training to decide which medicines are right for a specific patient. This information does not endorse any medicine as safe, effective, or approved for treating any patient or health condition. This is only a brief summary of general information about this medicine. It does NOT include all information about the possible uses, directions, warnings, precautions, interactions, adverse effects, or risks that may apply to this medicine. This information is not specific medical advice and does not replace information you receive from the healthcare provider. You must talk with the healthcare provider for complete information about the risks and benefits of using this medicine.