What is MS Contin?
MS Contin is:
- A strong prescription pain medicine that contains an opioid (narcotic) that is used to manage pain severe enough to require daily around-the-clock, long-term treatment with an opioid, when other pain treatments such as non-opioid pain medicines or immediate-release opioid medicines do not treat your pain well enough or you cannot tolerate them.
- A long-acting (extended-release) opioid pain medicine that can put you at risk for overdose and death. Even if you take your dose correctly as prescribed you are at risk for opioid addiction, abuse, and misuse that can lead to death.
- Not for use to treat pain that is not around-the-clock.
What is the most important information I should know about MS Contin?
- Get emergency help right away if you take too much MS Contin (overdose). When you first start taking MS Contin, when your dose is changed, or if you take too much (overdose), serious or life-threatening breathing problems that can lead to death may occur.
- Taking MS Contin with other opioid medicines, benzodiazepines, alcohol, or other central nervous system depressants (including street drugs) can cause severe drowsiness, decreased awareness, breathing problems, coma, and death.
- Never give anyone else your MS Contin. They could die from taking it. Selling or giving away MS Contin is against the law.
- Store MS Contin securely, out of sight and reach of children, and in a location not accessible by others, including visitors to the home.
Who should not take MS Contin?
Do not take MS Contin if you have:
- severe asthma, trouble breathing, or other lung problems.
- a bowel blockage or have narrowing of the stomach or intestines.
What should I tell my healthcare provider before taking MS Contin?
Before taking MS Contin, tell your healthcare provider if you have a history of:
- head injury, seizures
- problems urinating
- abuse of street or prescription drugs, alcohol addiction, or mental health problems.
- liver, kidney, thyroid problems
- pancreas or gallbladder problems
Tell your healthcare provider if you are:
- pregnant or planning to become pregnant. Prolonged use of MS Contin during pregnancy can cause withdrawal symptoms in your newborn baby that could be life-threatening if not recognized and treated.
- breastfeeding. Not recommended during treatment with MS Contin. It may harm your baby.
- taking prescription or over-the-counter medicines, vitamins, or herbal supplements. Taking MS Contin with certain other medicines can cause serious side effects.
How should I take MS Contin?
When taking MS Contin:
- Do not change your dose. Take MS Contin exactly as prescribed by your healthcare provider. Use the lowest dose possible for the shortest duration.
- Take your prescribed dose every 8 to 12 hours, as directed by your healthcare provider. Do not take more than your prescribed dose. If you miss a dose, take your next dose at the usual time.
- Swallow MS Contin whole. Do not cut, break, chew, crush, dissolve, snort, or inject MS Contin because this may cause you to overdose and die.
- Call your healthcare provider if the dose you are taking does not control your pain.
- Do not stop taking MS Contin without talking to your healthcare provider.
- Dispose of expired, unwanted, or unused MS Contin by promptly flushing down the toilet, if a drug take-back option is not readily available. Visit www.fda.gov/drugdisposal for additional information on disposal of unused medicines.
What should I avoid while taking MS Contin?
While taking MS Contin do not:
- Drive or operate heavy machinery, until you know how MS Contin affects you. MS Contin can make you sleepy, dizzy, or lightheaded.
- Drink alcohol or use prescription or over-the-counter medicines that contain alcohol. Using products containing alcohol during treatment with MS Contin may cause you to overdose and die.
What are the possible side effects of MS Contin?
The possible side effects of MS Contin are:
- Constipation, nausea, sleepiness, vomiting, tiredness, headache, dizziness, abdominal pain. Call your healthcare provider if you have any of these symptoms and they are severe.
Get emergency medical help if you have:
- trouble breathing, shortness of breath, fast heartbeat, chest pain, swelling of your face, tongue or throat, extreme drowsiness, light-headedness when changing positions, feeling faint, agitation, high body temperature, trouble walking, stiff muscles, or mental changes such as confusion.
These are not all the possible side effects of MS Contin. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
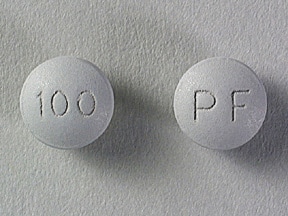
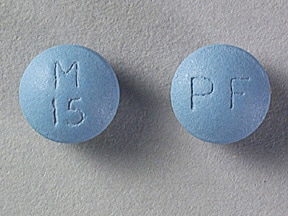
MS Contin Images
How should I store MS Contin?
Store at 25°C (77°F); excursions permitted between 15° to 30°C (59° to 86°F).
Store MS Contin securely and dispose of properly.
What are the ingredients in MS Contin?
Active ingredient: morphine sulfate
Inactive ingredient:
15mg: cetostearyl alcohol; hydroxyethyl cellulose; hypromellose, unspecified; magnesium stearate, polyethylene glycol, unspecified; talc; titanium dioxide; FD&C Blue No. 2; lactose, unspecified form; polysorbate 80.
30mg: cetostearyl alcohol; hydroxyethyl cellulose; hypromellose, unspecified; magnesium stearate; polyethylene glycol, unspecified; talc; titanium dioxide; D&C Red No. 7; FD&C Blue No. 1; lactose, unspecified form; polysorbate 80.
60mg: cetostearyl alcohol; hydroxyethyl cellulose; hypromellose, unspecified; magnesium stearate, polyethylene glycol, unspecified; talc; titanium dioxide; D&C Red No. 30; D&C yellow No. 10; hydroxypropyl cellulose, unspecified; lactose, unspecified form.
100mg: cetostearyl alcohol; hydroxyethyl cellulose; hypromellose, unspecified; magnesium stearate, polyethylene glycol, unspecified; talc; titanium dioxide; Ferrosoferric oxide.
200mg: cetostearyl alcohol; hydroxyethyl cellulose; hypromellose, unspecified; magnesium stearate, polyethylene glycol, unspecified; talc; titanium dioxide; D&C yellow No. 10; FD&C Blue No. 1; hydroxypropyl cellulose, unspecified.